Smart Pill Adherence & Signal Integrity Calculator
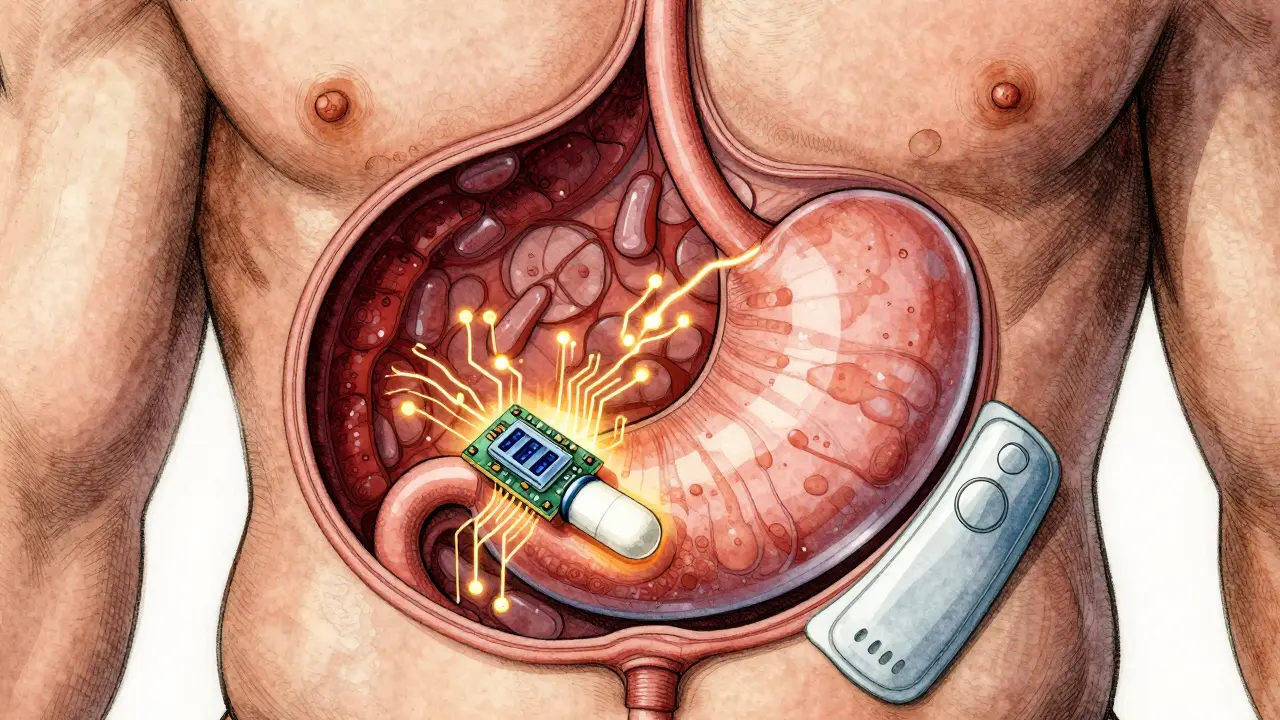
Sensor Power: Activated by gastric acid (~1-2V)
Transmission: Bluetooth Low Energy (BLE)
Security: AES Encryption
Why Smart Pills Are Changing Healthcare
Imagine swallowing a tablet that instantly tells your doctor you took it. That is exactly what Digital Pill Sensors are designed to do. These are not magic tablets; they are advanced drug-device combinations integrating traditional medication with microscopic monitoring systems. For patients managing chronic conditions like schizophrenia or HIV/AIDS, missing a dose can be dangerous. Historically, doctors rely on patient honesty or empty pill bottle counts, which often lead to incorrect assumptions. The World Health Organization notes that adherence rates for chronic diseases hover around 50%, meaning half the time, the necessary medicine simply isn’t taken. This gap creates massive costs and poorer health outcomes. Digital pill sensors aim to close this gap by verifying ingestion in real-time. Since the Food and Drug Administration (FDA) first approved a system involving aripiprazole for schizophrenia back in 2017, the technology has evolved. We have moved beyond simple tracking. Now, researchers are integrating these sensors to detect physiological changes, offering insights into side effects rather than just intake. If you or a loved one uses complex medication regimens, understanding how this technology works-and where its limits lie-is essential before considering adoption.
How the Sensor Detects Ingestion
The core of this system lies in the ingestible sensor itself. Think of it as a tiny circuit board embedded inside the pill capsule. It typically measures about 5 mm in diameter and 0.3 mm thick. When the pill reaches your stomach, gastric fluids activate a micro-battery. This process involves a copper-magnesium electrode system. Upon contact with stomach acid, an electrochemical reaction generates a small electrical charge, roughly 1 to 2 volts. This power-up moment triggers the device to broadcast a unique digital ID code. This transmission doesn’t happen directly to a cloud server. Instead, the signal travels via Bluetooth Low Energy (BLE) to a wearable patch usually stuck to the abdomen. The patch acts as a relay, receiving the ingestion event and timestamping it accurately. From there, it sends the data through a mobile app to a secure web portal. Without the patch nearby, the system cannot confirm the dose was taken. This architecture ensures data flows securely without exposing sensitive health information during transit. Protocols like Advanced Encryption Standard (AES) protect the signal between the body and the phone.
| Component | Specification | Function |
|---|---|---|
| Sensor Size | 5 mm x 0.3 mm | Fits inside standard capsules |
| Activation | Gastric fluid | Powers up only upon ingestion |
| Transmission | Bluetooth Low Energy | Sends signal to wearable patch |
| Data Security | AES Encryption | Protects transmitted data |
| Wearable Range | Several meters | Relays data to smartphone |
Clinical Benefits and Real-World Usage
Hospitals and research teams have deployed these tools across diverse areas. Mental health remains the biggest sector, accounting for nearly half of all implementations. Schizophrenia and bipolar disorder require strict adherence to prevent relapse. In a recent trial involving 157 participants, adherence jumped from 62% to 84% using digital monitoring. However, benefits extend beyond mental health. Tuberculosis treatment programs recently adopted similar technology to prevent antibiotic resistance caused by skipped doses. Beyond simple logging, some advanced versions are starting to monitor side effects. For instance, systems like the IntelliCap developed by Philips Research can track gastrointestinal temperature and pH levels. This data helps physicians distinguish between a missed dose and a genuine adverse reaction. If a patient stops taking medication due to nausea, the system records the drop in activity alongside physiological stress markers. Doctors get a clearer picture of why a regimen fails. It shifts the conversation from "Did you take your meds?" to "Is the medication tolerating well enough to continue?" Companies like etectRx and Medtronic (who acquired Proteus Digital Health) are leading this space. Their platforms connect patient data to clinician dashboards, automating reports. A physician logs in and sees a calendar of adherence events. Red flags pop up automatically when gaps appear. This automation reduces administrative burden, allowing care teams to intervene sooner.
Privacy Concerns and Data Protection
While the health benefits are significant, the trade-off involves personal privacy. You are essentially wearing a tracker that broadcasts intimate health habits. The Electronic Frontier Foundation has highlighted risks regarding insurance companies or employers accessing this sensitive history. Even with laws like the Health Insurance Portability and Accountability Act (HIPAA) providing safeguards, many patients worry about long-term data storage. Surveys show that over two-thirds of potential users cite privacy as their primary hesitation. Patients often feel monitored. In online forums, some describe the sensation as a loss of autonomy. One user noted feeling like their psychiatrist was watching them swallow every pill. To address this, developers implement local encryption. The raw biometric data stays on the secure server, accessible only to authorized providers. Additionally, patients control the connection settings. They can disconnect the app or remove the patch whenever they wish. Yet, the knowledge that the doctor “sees” everything can create pressure to perform compliance rather than genuine recovery. Regulatory frameworks are catching up. The FDA requires separate approvals for the drug component and the sensor device. This dual-review process ensures safety standards are met for both biological and electronic aspects. However, data governance varies by state. Fourteen U.S. states have enacted specific digital health privacy laws, creating a patchwork of regulations for companies operating nationally. Users must read consent forms carefully to understand who holds the rights to their ingestion data.

Limitations and Technical Challenges
Despite the hype, the technology isn't perfect. Hardware limitations persist. The wearable patch battery typically lasts about 72 hours before needing a change or recharge. Signal interference is another hurdle. Approximately 12 to 15% of transmissions fail in real-world testing due to body mass index or interference from other electronics. Patients with a BMI over 35 see higher failure rates, sometimes reaching 18%. Fat tissue can absorb signals, preventing the patch from picking up the pill's signal. Another major constraint is cost. Implementation requires training staff for 3 to 5 hours to interpret data correctly. Setup takes time-roughly 20 minutes per patient. Reimbursement models lag behind innovation. Most insurers currently classify these as experimental or administrative tools, rarely covering the full cost of the hardware and software subscription. The Congressional Budget Office estimates reimbursement structures would need to increase by 15-20% to support widespread adoption without raising healthcare costs. There is also the "therapeutic misconception." Some patients assume the sensor makes the drug work better. Dr. Michelle Mello warned that people might believe the monitoring provides therapeutic benefit. The sensor tracks behavior; it does not cure illness. Understanding this distinction prevents false expectations about treatment efficacy.
Future Trends and Predictive AI
The next frontier involves artificial intelligence predicting adherence lapses. Partnerships like the one between etectRx and IBM Watson Health aim to analyze historical patterns. Algorithms predict missed doses with over 80% accuracy based on contextual factors like travel or sleep disruptions. By 2026, projections suggest that 60% of systems will integrate side-effect detection capabilities natively. We are also seeing multi-parameter sensing expand. Sensors now measure pH within ±0.3 units and temperature within ±0.5°C. This granular data allows for remote monitoring of inflammation or infection signs. If a patient develops fever, the patch might flag it before they feel symptomatic. Integration with broader health platforms allows these devices to talk to wearables like smartwatches, consolidating heart rate and step count data alongside adherence metrics. Experts predict digital pills will become standard for high-risk regimens within five years. However, routine medications may not justify the cost yet. The transition is gradual, balancing innovation with practical utility.
Are digital pills safe to swallow?
Yes, the sensors are biocompatible and designed to pass safely through the digestive tract. They are typically made from food-grade materials and dissolve or exit naturally after transmitting data.
Do I need to wear a patch every day?
Yes, the wearable patch is required to receive the signal from the pill. Most patches last 72 hours and need replacement three times a week.
Can doctors see my data in real time?
Data is usually batched and uploaded regularly. While technically possible to stream in real-time, most systems send updates periodically to preserve battery life and reduce server load.
Does health insurance cover these systems?
Coverage varies significantly. Many plans still categorize them as research tools, though this is changing as adoption grows in mental health and transplantology.
What happens if the signal fails?
If the patch misses the signal, the record shows a gap. Patients should try moving the patch closer to the stomach area. Systematic failures indicate a hardware issue requiring support intervention.






Mark Zhang
It is truly inspiring to see technology moving towards such personalized health monitoring solutions. Many patients struggle silently with adherence without anyone noticing the pattern. Having an objective measure could genuinely save lives by catching gaps early before relapse occurs. The mental health sector especially stands to gain massive improvements with these tools integrated into care plans. We should encourage wider adoption while keeping ethical boundaries clear for patient consent.
Vicki Marinker
One must question the efficacy of relying on hardware when human error remains so prevalent in daily life. The cost implications alone suggest this solution targets those who can afford expensive subscriptions rather than genuine public health improvement.
Rachelle Z
Oh! The usual privacy naysayer showing up! :) Everyone knows they want free healthcare too! But seriously, who wants to track their own health like a lab rat anyway!??
Aysha Hind
This digital ingestion tracking feels suspiciously aligned with recent agendas regarding bio-data monetization. Pharmaceutical giants would love to sell our compliance history to insurance carriers for profit margins. Think about the data trail left every time you swallow a capsule designed to broadcast your location and habits. It reminds one of older surveillance tactics wrapped in shiny medical packaging for mass appeal. They claim it saves lives but ignore the erosion of bodily autonomy required for such constant monitoring systems. A glitched system could effectively create a criminal record for missed doses under upcoming healthcare statutes. The encryption standards mentioned seem weak against sophisticated state-level actors interested in population control metrics. Patients are already feeling monitored pressure which destroys trust in the therapeutic relationship itself. One might consider social support networks better than chips when financial incentives drive development away from genuine help. It feels like we are entering a dark age of mandated electronic supervision disguised as voluntary medical enhancement. Big Pharma profits soar when adherence is guaranteed through fear of detection rather than genuine wellness goals. We should scrutinize the patents held by companies owning these sensor technologies before integration becomes standard policy. History shows us that convenience often paves the road toward total loss of personal freedom in sensitive areas. The patch replacement schedule ensures constant contact points with your physical skin that could collect more than just pill signals. It remains unclear whether the gastric activation triggers functions beyond simple battery power during digestion processes.
Hudson Nascimento Santos
The philosophical tension between safety and liberty is always present when new medical surveillance enters society.
sophia alex
You speak of liberty yet ignore the reality of privilege afforded to those paying for such elite treatment options! :D Only the wealthy need worry about such grand philosophical concepts while the poor simply survive.
Lawrence Rimmer
The machine always wins eventually.
The Charlotte Moms Blog
What a nihilistic statement!!! Does nothing matter if machines rule everything?! You sound like a failed writer trying to be deep!!!
Hope Azzaratta-Rubyhawk
Progress requires bold statements despite the cynicism that slows necessary medical advancements forward.
simran kaur
The idea of trusting these sensors completely without knowing who holds the master keys is reckless beyond belief.